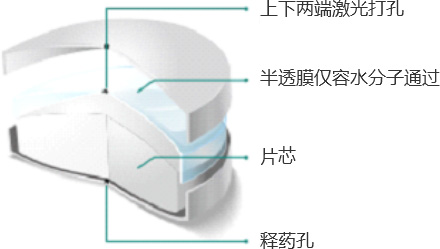
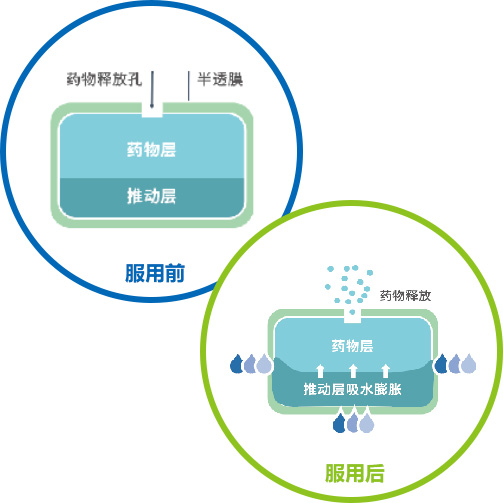
(1) Osmotic Pump Technology
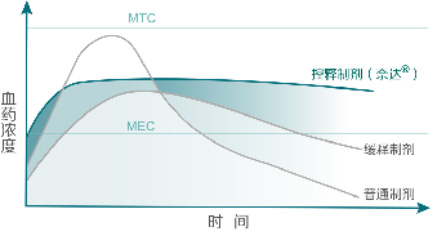
Osmotic pump coating technology uses osmotic pressure regulators to produce a constant osmotic effect, releasing the drug at a steady rate over 24 hours. This is its core advantage over conventional tablets or matrix sustained-release tablets.Clinically, osmotic pump drugs deliver more stable plasma drug concentrations in vivo, longer-lasting therapeutic effects, and avoid plasma drug peak-trough fluctuations, thus improving safety. They also provide better patient compliance: once-daily dosing reduces the risk of missed doses or taking multiple tablets at once.
Qingdao Baheal Pharmaceutical has mastered mature osmotic pump coating technology, which is subdivided into single-chamber and dual-chamber osmotic pumps. The company has independently developed multiple products that have been successfully launched, manufactured and commercially marketed.Single-chamber osmotic pump products include Metformin Hydrochloride Sustained-Release Tablets (III) and Carbamazepine Sustained-Release Tablets (II) (contract manufacturing). Dual-chamber osmotic pump products include Nifedipine Extended-Release Tablets and Glipizide Extended-Release Tablets (contract manufacturing).Product R&D follows the Quality by Design (QbD) principle. Systematic scientific evaluation and research are performed on Critical Material Attributes (CMAs, including osmotic pump functional polymers) and Critical Process Parameters (CPPs) using Design of Experiments (DOE). This ensures stable, controllable quality, reproducible manufacturing processes, and continuous quality improvement throughout the product lifecycle.
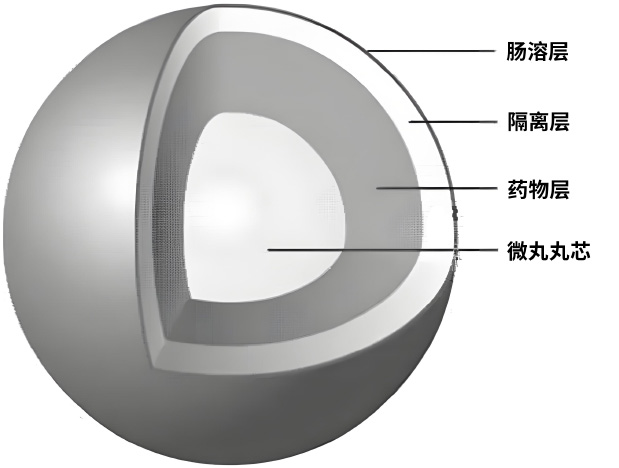
(2) Pellet Sustained-Release Coating Technology
Pellet sustained-release coating technology enables controlled drug release, intestinal-specific or targeted delivery, pulsed delivery, timed or programmed administration, and taste masking for improved palatability, significantly reducing the drug’s toxic and side effects.This technology can be formulated into capsules, tablets, dry suspensions and other dosage forms.For example, Memantine Hydrochloride Sustained-Release Capsules provide once-daily controlled delivery with 24-hour efficacy. Duloxetine Hydrochloride Enteric-Coated Capsules achieve intestinal targeted delivery, avoiding gastric irritation and preventing degradation by gastric acid, thereby enhancing drug stability and ensuring safety.